The rate equation, often called the rate law, is the empirical relationship that links the speed of a chemical reaction to the concentrations of reactants and a proportionality constant. It is a central concept in chemical kinetics because it summarizes how changing concentrations, temperature, pressure or catalysts affects how rapidly products form. The law must be established experimentally for most reactions, although simple mechanisms can sometimes predict its form. Rate equation and reaction rate are closely related terms used across physical chemistry, biochemistry and industrial processes.
Basic form and terms
For a reaction written generically as aA + bB → products, the usual algebraic form is r = k [A]^x [B]^y, where r is the instantaneous rate, [A] and [B] are species concentrations, k is the rate constant, and x and y are the reaction orders with respect to A and B. The overall order is the sum x + y. The rate constant k carries units that depend on this overall order and is sensitive to conditions such as temperature and catalysts. The exponents x and y are determined by the reaction mechanism rather than the stoichiometric coefficients in general; see concentration and rate-determining step concepts.
Orders, mechanisms and experimental determination
Reaction orders (zero, first, second, fractional, etc.) are found by measuring how the rate changes when concentrations are varied. A first-order process satisfies r = k[A], producing an exponential decay of [A] with time; integration gives ln[A] = −kt + ln[A]0 so a plot of ln[A] versus t is linear with slope −k. More complex mechanisms and intermediate steps can produce different apparent orders; for example, if one reactant remains in large excess its concentration can be treated as constant and the observed rate may appear first order (a pseudo-first-order condition). The concepts of reaction mechanism and transition state often guide interpretation.
Mathematical character and condition dependence
Rate equations are differential expressions: they relate the time derivative of concentration to a function of concentrations, and are integrated to predict concentration vs. time profiles. Because k commonly depends on temperature (Arrhenius behavior) and can be influenced by pressure, solvent, ionic strength or catalysts, kinetic parameters are typically reported with the conditions used to measure them. For more on these mathematical aspects see entries on temperature effects, pressure dependence, the underlying differential equation form, and techniques for integration.
Applications and examples
- Laboratory kinetics: determining mechanisms by comparing observed orders with proposed elementary steps.
- Enzymology: enzyme-catalyzed reactions use modified rate laws (e.g., Michaelis–Menten) to describe substrate dependence and saturation.
- Industrial reactors: design and scale-up rely on integrated rate laws to size reactors and predict yields over time.
- Environmental chemistry and atmospheric models: rate expressions predict pollutant formation and decay under varying conditions.
Practical analysis often combines experimental data, mechanistic hypotheses and kinetic modeling. Simple integrated laws (zero, first, second order) serve as diagnostics, while numerical integration of coupled rate equations is used for complex networks.
Notable distinctions and cautions
Important distinctions include the difference between molecularity (an attribute of an elementary step) and reaction order (an empirical exponent), and the fact that stoichiometric coefficients do not always equal orders. Reaction orders can be non-integer or change with conditions when mechanisms shift. Kinetic data should always be reported alongside experimental conditions and the method used to determine orders and rate constants, because those parameters are not intrinsic constants in the same sense as molecular masses. Careful interpretation links rate laws to plausible microscopic steps and to thermodynamic constraints without over-interpreting limited data.
For further reading, introductory texts and specialized reviews discuss experimental techniques, integrated forms for common orders, and how to extract mechanistic information from kinetic measurements. Additional resources and detailed examples are available through linked references above.





![{\displaystyle f_{i}(\{a_{i}\})=\sum _{r}|\nu _{i,r}|\left[-k_{\text{-,r}}\prod _{{\text{Reaktanden j}},r}a_{r,j}^{|\nu _{r,j}|}+k_{\text{+,r}}\prod _{{\text{Produkte j}},r}a_{r,j}^{|\nu _{r,j}|}\right].}](https://www.alegsaonline.com/image/b80db270a3bf31f7c4be6600b060091e05d50b94.svg)










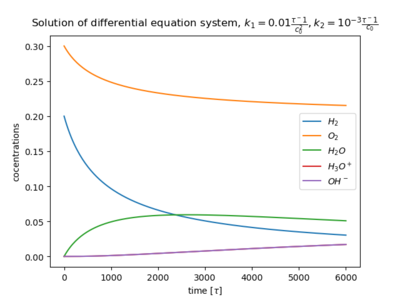
![{\displaystyle {\frac {\mathrm {d} }{\mathrm {d} t}}\mathrm {[H_{2}]} =-2k_{1}\mathrm {[H_{2}]^{2}[O_{2}]} }](https://www.alegsaonline.com/image/2c13665e60d079c9c00548aa0321ec4bab0f19bf.svg)
![{\displaystyle {\frac {\mathrm {d} }{\mathrm {d} t}}\mathrm {[O_{2}]} =-k_{1}\mathrm {[H_{2}]^{2}[O_{2}]} }](https://www.alegsaonline.com/image/b250606928c4804538db7c25ae171f3814b10de8.svg)
![{\displaystyle {\frac {\mathrm {d} }{\mathrm {d} t}}\mathrm {[H_{2}O]} =+2k_{1}\mathrm {[H_{2}]^{2}[O_{2}]} -2k_{2}\mathrm {[H_{2}O]^{2}} }](https://www.alegsaonline.com/image/2ff8e0597eaea466b1d3a2394ae0d6e191450de3.svg)
![{\displaystyle {\frac {\mathrm {d} }{\mathrm {d} t}}\mathrm {[H_{3}O^{+}]} =+k_{2}\mathrm {[H_{2}O]^{2}} }](https://www.alegsaonline.com/image/008993d5ea22cfbbff2fadbb68eb25d19621909a.svg)
![{\displaystyle {\frac {\mathrm {d} }{\mathrm {d} t}}\mathrm {[OH^{-}]} =+k_{2}\mathrm {[H_{2}O]^{2}} }](https://www.alegsaonline.com/image/56a36873b3b7c5d6c84f6c7fb5058e45703f27e4.svg)
![{\displaystyle c_{1}\equiv \mathrm {[H_{2}]} ,\;\;c_{2}\equiv \mathrm {[O_{2}]} ,\;\;c_{3}\equiv \mathrm {[H_{2}O]} ,\;\;c_{4}\equiv \mathrm {[H_{3}O^{+}]} ,\;\;c_{5}\equiv \mathrm {[OH^{-}]} }](https://www.alegsaonline.com/image/a6939591f470059cb3714a724ce62a23d23fee9a.svg)