Palladium was unwittingly used as a component of platinum alloys by the pre-Columbian Indians of Ecuador and Colombia. A number of platinum jewels were found there containing about 85% platinum, 7% iron and 4.6% of a mixture of the platinum metals palladium, rhodium and iridium, and copper.
William Hyde Wollaston discovered palladium in a South American platinum ore in 1802. He had dissolved the ore in aqua regia and then neutralized the solution with sodium hydroxide. He then precipitated the platinum with ammonium chloride as ammonium hexachloroplatinate and separated it. By adding mercury cyanide to the remaining solution Wollaston obtained palladium cyanide, from which he obtained metallic palladium by heating.
As early as 1866, Thomas Graham noted the amazing storage capacity of finely divided palladium for hydrogen, which can hold about 900 times its own volume of hydrogen gas at room temperature and atmospheric pressure. This led to the assumption that hydrogen was a very volatile metal and that the palladium with the trapped hydrogen was an alloy of this volatile metal.
Francis Clifford Phillips, a US chemist, discovered the stoichiometric oxidation of ethene to acetaldehyde using palladium(II) chloride in 1894 when he was investigating the oxidation of naturally occurring hydrocarbons. Towards the end of the 1950s, Wacker-Chemie converted the stoichiometric reaction discovered by Phillips into a catalytic variant in the Wacker-Hoechst process. In the process, which produced millions of tons of acetaldehyde and its downstream product acetic acid per year, the chemical industry used a palladium catalyst in a large-scale application for the first time. It was also the first large-scale homogeneous catalytic process.
From the end of the 1960s, palladium salts were used for coupling reactions. This led to the development of reactions that are important for organic chemistry, such as the Heck reaction, the Stille coupling, the Suzuki coupling and the Negishi coupling. Three of the researchers involved, Richard F. Heck, Ei-ichi Negishi and Akira Suzuki, were awarded the Nobel Prize in Chemistry in 2010.
Electrochemical adsorption experiments in 1989 by Martin Fleischmann and Stanley Pons with the palladium-deuterium system became known as "cold fusion" and hit the headlines worldwide. The supposed "cold fusion" of deuterium triggered by palladium was considered a scientific sensation for a short time with the hope that this could provide a virtually inexhaustible source of energy.
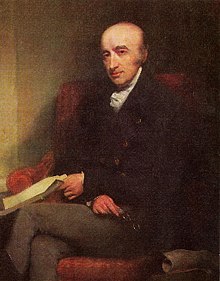


.PNG)