Overview
Nitrogen is a colorless, odorless diatomic gas at standard conditions and is classified as a nonmetal. It has the chemical symbol N and atomic number 7. Molecular nitrogen (N2), in which two nitrogen atoms are joined by a strong triple bond, is the dominant form in Earth's atmosphere and plays a central role in both natural processes and human industry.
Physical and chemical properties
The nitrogen atom contains seven protons in its nucleus and typically seven neutrons in the most common isotope. Its electrons arrange to give five valence electrons, which govern its chemistry. The N≡N triple bond in molecular nitrogen is unusually strong, making N2 relatively inert at ordinary temperatures. Nitrogen forms a wide range of compounds, from reduced species such as ammonia to highly oxidized oxides (NO, NO2) and nitrates.
Occurrence, cycle and biological importance
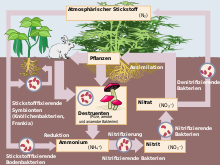
About 78 percent of the atmosphere is molecular nitrogen, which provides a vast reservoir of nitrogen but is not directly usable by most organisms. Conversion of N2 into biologically available forms is achieved by biological nitrogen fixation, industrial processes and lightning. Fixed nitrogen is incorporated into amino acids, proteins, nucleic acids and chlorophyll, so the nitrogen cycle is fundamental to plant growth, ecosystems and food production. See more about atmospheric composition at atmosphere resources.
Production and major uses
Industrial fixation of nitrogen, most famously by the Haber–Bosch process, combines nitrogen with hydrogen to produce ammonia, the basis for most synthetic fertilizers. Ammonia and its derivatives support modern agriculture and thus global food supply. Nitrogen and its compounds also have uses in refrigeration (liquid nitrogen), chemical synthesis, explosives (nitro compounds), pharmaceuticals, and as an inert atmosphere for sensitive manufacturing.
History and notable facts
Nitrogen was identified as a distinct component of air in the late 18th century. Its relative chemical inertness arises from the strong triple bond in N2, which must be broken for the element to react. Two stable isotopes, one much more abundant than the other, are used in scientific studies and tracing of biological and geological processes. For quick reference to its basic atomic information, consult a summary of its atomic structure and common properties.
Key points
- Essential element for life: component of amino acids and nucleic acids.
- Atmospheric reservoir: N2 comprises roughly three quarters of Earth's air.
- Industrial importance: ammonia production underpins modern fertilizers and food production.
- Environmental considerations: excess fixed nitrogen can cause eutrophication and other ecological impacts.