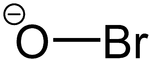Hypobromite is an ion. Its chemical formula is BrO-. It contains bromine in its +1 oxidation state. It is an oxidizing agent. It is similar to hypochlorite. Hypobromites are the salts of hypobromous acid. Hypobromite is a disinfectant. Some white blood cells oxidize bromide to hypobromite with peroxide and use the hypobromite to kill germs. Bromides can be added to pools along with chlorine to make hypobromite, which disinfects the pools.
Hypobromites are made by dissolving bromine in a basic solution. Hypobromite disproportionates to bromate and bromide when heated.