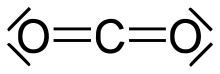Overview
Group 14, often called the carbon group, occupies one vertical column of the periodic table and contains five stable, commonly discussed elements. Members share a valence shell configuration with four outer electrons, which gives them characteristic bonding patterns and multiple oxidation states. The group is important in chemistry, materials science and technology; see the periodic table for its location.
Electronic structure and common oxidation states
Atoms in this group have the ns2np2 outer configuration, which allows formation of up to four covalent bonds or electron-pair sharing. They frequently exhibit the +4 oxidation state, but the heavier members more readily stabilize a +2 state due to increasing inert-pair effects. Carbon and its derivatives can also form negatively charged species (carbanions) under many conditions; for more on valence and bonding patterns see valence electrons and discussions of covalent bonding at bonding concepts. The tendency to form carbanions and other anionic species is most pronounced for carbon; see carbanions.
Members of the group
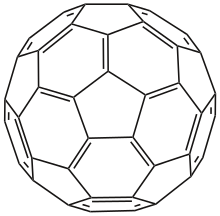
Carbon — a nonmetal with numerous allotropes (graphite, diamond, fullerenes) and central to organic chemistry. Carbon chemistry underpins life and fuels materials innovation.
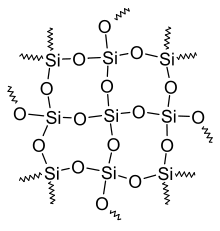
Silicon — a metalloid used widely in semiconductors and glass manufacture; it bridges nonmetal and metal behavior. Silicon is the backbone of the electronics industry.
Germanium — a semiconductor with historical and niche uses in electronics and infrared optics; it behaves between silicon and metals. Germanium is less abundant than silicon.
Tin — a post-transition metal used in alloys (bronze, solder) and coatings; it shows both +4 and +2 oxidation states. Learn more at tin.
Lead — a dense, soft metal historically used in pipes, paints and batteries; its toxicity has reduced many uses. Lead commonly shows +2 and +4 states. See lead.
Physical trends, allotropy and the superheavy neighbor
Across the group there is a steady shift from nonmetallic to metallic behavior: carbon is a true nonmetal, silicon and germanium are semimetals (metalloids) with semiconducting properties, and tin and lead are classified as poor metals. Heavier elements show larger atomic radii, lower ionization energies and increased metallic character. Superheavy elements homologous to Group 14 have been synthesized briefly and are short-lived; their properties are still under investigation in advanced laboratories and theoretical work. For typical oxidation behavior see oxidation states.
Uses, occurrences and notable facts
- Carbon: foundation of organic chemistry, fuels, polymers, nanomaterials and biomolecules.
- Silicon and germanium: core materials for microelectronics and photovoltaic devices.
- Tin: soldering, plating and corrosion-resistant alloys.
- Lead: historically widespread but now constrained by health regulations; still used in some batteries and radiation shielding.
Group 14 elements illustrate how a single valence electron configuration can produce a wide variety of chemical and physical behaviors. Their study links fundamental atomic theory with practical technologies from structural materials to advanced electronics.