During solidification, numerous physical properties change abruptly. Almost all physical properties change with the temperature of a body. As long as the body does not change its state of aggregation, these properties usually do not change abruptly, but continuously and very slowly. Changes in volume, density and solubility are of particular importance.
Volume and density
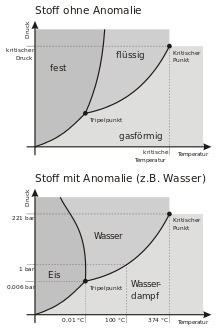
Most substances reduce their volume during solidification and thus increase their density (mass per volume) because of the constant mass. This phenomenon is called solidification shrinkage. Water, on the other hand, expands during solidification. This is why ice floats on liquid water, while most other substances sink in the melt. The expansion when water freezes can cause glass bottles filled with water to burst. In winter, water seeps into small cracks in pavement and rock, expanding and thus enlarging these cracks, which is called frost heave. Thermal expansion, on the other hand, is related to temperature and not to the state of aggregation.
In the foundry, the solidification shrinkage can cause damage to the castings. Especially with complex shaped workpieces, the reduction of the casting is blocked by the mould, which can lead to cracks. These are called hot cracks and can also occur during welding. In order to keep shrinkage as low as possible, silicon is often added to cast alloys, as it expands during solidification and can thus partially compensate for shrinkage.
Solubility
Solubility also changes greatly with solidification. In general, a substance can dissolve less and less of another substance as it cools. During solidification, however, the solubility drops sharply. If impurities are dissolved in a substance, they can be removed by recrystallization or the electroslag remelting process, since the impurities remain primarily in the area that has not yet solidified. In the foundry, gases are often also dissolved in the melt. When the melt cools rapidly, these gases do not have sufficient time to escape from the melt and then remain in the casting where they form bubbles and pores, which reduces the strength of the castings. When these castings are subsequently welded, the gases in the pores expand greatly and can damage the workpiece.