First work
The first work on the hydrogenation of carbon monoxide with hydrogen was carried out by Paul Sabatier and Jean Baptiste Senderens in 1902. Using nickel and cobalt catalysts, they obtained water and methane as the main products at atmospheric pressure and temperatures between 200 and 300 °C. The process would have increased the calorific value of town gas and avoided the toxic carbon monoxide it contained, but did not catch on due to cost. Sabatier received the Nobel Prize in Chemistry in 1912 for his work on hydrogenation.
In 1913, BASF filed a patent describing the production of saturated and unsaturated aliphatic hydrocarbons by hydrogenation of carbon monoxide at a pressure of about 100 bar and temperatures of 400 °C using nickel, cobalt, zinc and other metals or their oxides as catalysts.
Franz Fischer, founding director of the Kaiser Wilhelm Institute for Coal Research in Mülheim an der Ruhr, which was established in 1912 and whose maintenance was largely funded by the coal industry, focused his research in the early 1920s on the utilization of coke oven gas. This was a mixture of hydrogen, methane, nitrogen and carbon monoxide that was produced during the coking of coal and was available to heavy industry in surplus.
In 1921, together with Hans Tropsch, he developed a process based on alkalized iron-containing catalysts for the production of mixtures of oxygen-containing compounds from synthesis gas, the so-called synthol process. At a pressure of 100 to 150 bar and a temperature of about 400 °C, they obtained mixtures of aldehydes, ketones, carboxylic acid esters, alcohols and carboxylic acids, so-called synthol.
In the following year, the chemist Matthias Pier succeeded in producing methanol on an industrial scale by hydrogenating carbon monoxide in a high-pressure process on alkali-free zinc oxide-chromium oxide catalysts. Since the presence of iron led to methane formation, the reactors were lined with copper. The use of alkalized contacts yielded higher alcohols, especially isobutanol. With these successes, BASF focused its carbon monoxide hydrogenation research on methanol production and isobutyl oil synthesis.
Work at the Kaiser Wilhelm Institute for Coal Research
The shortage of petroleum and the associated bottleneck in fuel supplies during the First World War triggered a politically and militarily motivated search in the German Empire for alternative solutions, primarily based on domestic coal.
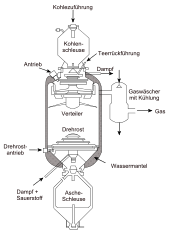
Fischer initially commissioned Otto Roelen, who had already been part of the Mülheim Institute since 1921 as Tropsch's assistant, to further investigate the synthol processS from 1924. After the oxygen-containing mixtures produced with it had proved unsuitable for use as motor fuel, Tropsch changed the reaction conditions and carried out the experiments under normal pressure. Although only low yields were achieved at the beginning, the work of Fischer, Tropsch and Roelen eventually led to the technical production of aliphatic hydrocarbons using cobalt or iron catalysts.
Large-scale process
During the Second World War, FT synthesis gained (wartime) economic importance in Germany. With it, the demand for liquid fuels, so-called Kogasin, as the synthesis product was called after its production sequence of coke, gas, gasoline, could be covered from domestic coal. By the end of the Second World War, the chemical industry had built a total of nine plants using FT synthesis, with a capacity of 0.6 million t/a.
Since Fischer-Tropsch fuels had a low octane rating, the National Socialist economic management built more hydrogenation plants instead, using the Bergius-Pier process, which provided a higher-grade motor gasoline; paraffin slack wax became difficult to obtain. It was not until 1940 that the Fischer-Tropsch plants significantly increased their output, resulting in a good supply of slack wax. At Imhausen's instigation, Wilhelm Keppler, the Commissioner for Economic Affairs, had the Fischer-Tropsch plants converted to the medium-pressure process, which provided a higher yield of paraffin slack wax.
A process developed by Arthur Imhausen used the higher molecular weight fraction, paraffin slack wax, for fatty acid synthesis by paraffin oxidation. The fatty acids were used as a raw material in the Märkische Seifenindustrie company, but from 1939 they were also used to produce a synthetic edible fat.
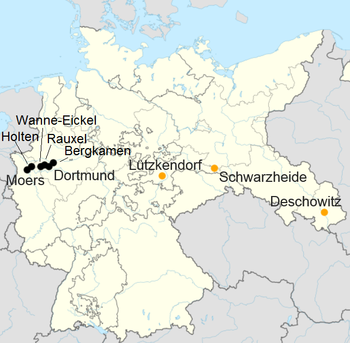
| Production of synthetic fuel by the Fischer-Tropsch process |
| Location | Operator | Input material | Production capacity
in 1943/44
in tonnes
per year |
| Holten | Ruhrchemie | Hard coal | 60.000 |
| Rauxel | Union Victor | Hard coal | 40.000 |
| Wanne-Eickel | Krupp | Hard coal | 55.000 |
| Bergkamen | Essen coal mines | Hard coal | 85.000 |
| Dortmund | Hoesch | Hard coal | 55.000 |
| Moers | Rheinpreussen | Hard coal | 75.000 |
| Schwarzheide | BRABAG | Lignite | 180.000 |
| Lützkendorf | Mitteldeutsche Treibstoff- und Oelwerke A.G. | Lignite | 30.000 |
| Deschowitz | Schaffgotsch Petrol Ltd. | Lignite | 40.000 |
| Total of all FT systems | | 620.000 |
Postwar
FT synthesis products were never competitive with petroleum-based fuels, so the industry almost completely abandoned the process after the war. The plants were dismantled in accordance with the Washington Decision of the Western powers. After the oil crisis, however, research was resumed in the 1970s and a pilot plant was built in Bottrop. This was already shut down again at the end of the 1980s, as production was only profitable when the price of petrol was above 2.30 German marks.
Procedure of Sasol
In the Republic of South Africa, which also had sufficient coal resources and had to import crude oil, South Africa's first modern coal-to-liquid (CtL) plant was commissioned in 1955 for political reasons. It was built by the specially founded Suid Afrikaanse Steenkool en Olie (Sasol) with the participation of the German Lurgi AG. The pilot plant Sasol 1 was designed for about 6,000 barrels of fuel per day. From 1980 onwards, capacities were significantly expanded due to South Africa's political development.
Thus, in 1980 and 1982, Sasol commissioned Sasol II and Sasol III. This provided a total capacity of 104,000 barrels/day. With the political opening, the programme was extended to natural gas as a feedstock source, and in 1995 and 1998 Sasol created further capacity for 124,000 barrels/day of CtL and GtL (gas-to-liquid) fuel. Since hard coal can be extracted relatively cheaply from opencast mines, the country was still meeting about 40 % of its fuel needs from coal gasoline in 2006.
Sasol became the world market leader in XtL technologies through its South African developments and built a modern GtL plant in Qatar in 2006 with a capacity of 34,000 barrels/day. This is a high-temperature process with process temperatures of 350 °C (synthol and advanced synthol) that produces gasoline and alkenes as platform chemicals. Together with Foster Wheeler, Sasol also planned a Fischer-Tropsch plant in China. This plant, with an annual capacity of 60,000 barrels, uses a low-temperature process at 250 °C and is used to produce diesel fuel and waxes.
Raw materials turnaround
In 1993, the oil company Royal Dutch Shell commissioned its first GtL plant. The plant in Bintulu in Malaysia has a capacity of 12,000 barrels/day and is operated using a specially developed Fischer-Tropsch process, the Shell Middle Distillate Synthesis (SMDS process). Together, Shell and Sasol aim to build further GtL capacity of around 60,000 barrels GtL/day.
The USA has large coal seams lying close to the surface that can be relatively easily extracted by opencast mining. In view of rising fuel prices and simultaneously high demand, the United States Air Force tested a Boeing B-52H on 19 September 2006 at Edwards Air Force Base with a 50:50 mixture of ordinary JP-8 fuel and fuel derived synthetically from coal. The test flight was intended to clarify how this fuel would perform in practice and whether it could be reliably operated economically. An accompanying research project concluded that Fischer-Tropsch fuels offer an alternative source of JP-8 for the US Air Force.
In the course of the raw material turnaround, biofuels in particular moved into the focus of fuel production. In this context, Fischer-Tropsch synthesis received renewed interest from research and development. Although biomass-to-liquid fuels are being promoted as second-generation biofuels, especially in Europe, no BtL production has yet come on stream. Individual pilot projects have been initiated, and the now insolvent Choren Industries wanted to produce the BtL fuel they call SunFuel and SunDiesel at a plant in Freiberg, Saxony.